The landscape of cancer immunotherapy has been irrevocably altered by the advent of mRNA technology. Following the unprecedented success of prophylactic mRNA vaccines on a global scale, the scientific community has rapidly pivoted its focus toward an even more complex challenge: therapeutic cancer vaccines. At the heart of this frontier lies mRNA vaccine design, a highly sophisticated process that seeks to turn the body’s own immune system against malignant cells.
However, success in this arena is not merely about synthesizing RNA. It requires solving a complex equation with two primary, interlocking variables: identifying the optimal biological targets and ensuring the therapeutic payload reaches its cellular destination effectively. In this comprehensive guide, we will explore how the critical pillars of neoantigen prioritization and mRNA deliverability intersect to shape the future of personalized cancer immunotherapy.
The Fundamentals of Oncology mRNA Vaccine Design
Therapeutic cancer vaccines operate on a fundamentally different premise than traditional prophylactic vaccines. Instead of preventing a viral infection, they are designed to treat an existing malignancy by training the patient’s immune system to recognize and eliminate tumor cells. The cornerstone of effective mRNA vaccine design lies in its inherent modularity. An mRNA vaccine acts as a set of genetic instructions, directing the patient’s own cellular machinery—specifically Antigen-Presenting Cells (APCs)—to produce specific tumor-associated antigens or mutated proteins.
The advantages of utilizing mRNA in this context are manifold. First, it is transient and non-integrating, entirely mitigating the risk of insertional mutagenesis associated with DNA-based viral vectors. Second, the manufacturing process is highly scalable and relatively agnostic to the encoded sequence, which is the foundational requirement for truly personalized medicine. Once the target sequence is identified, the production timeline can be compressed from months to mere weeks. However, maximizing this potential requires a delicate balance of molecular biology, bioinformatics, and materials science to ensure the synthetic mRNA is stable, highly translatable, and precisely targeted.
The Bottleneck: The Complexity of Neoantigen Prioritization
The defining feature of modern oncology mRNA vaccine design is its reliance on neoantigens. Unlike Tumor-Associated Antigens (TAAs), which are expressed on normal healthy cells at lower levels and often induce immune tolerance, neoantigens are entirely unique to the tumor. They arise from somatic mutations within the cancer genome, making them “non-self” to the immune system and highly immunogenic.
While a typical tumor may harbor hundreds or even thousands of mutations, only a tiny fraction of these will produce viable neoantigens capable of triggering a robust, tumor-killing T-cell response. Identifying this needle in the haystack is where neoantigen prioritization becomes the critical bottleneck. It is a highly demanding computational challenge.
The process begins with Next-Generation Sequencing (NGS) of the tumor DNA and RNA, alongside matched healthy tissue, to identify tumor-specific somatic mutations. Once mutant peptide sequences are generated in silico, bioinformatics algorithms must predict their affinity for the patient’s specific Human Leukocyte Antigen (HLA) alleles. But HLA binding is only the very first step.
Effective neoantigen prioritization must also evaluate a cascade of biological events:
-
Proteasomal Cleavage: Will the cell’s machinery cut the protein into the correct peptide fragments?
-
TAP Transport: Will these fragments be successfully transported into the endoplasmic reticulum?
-
T-Cell Receptor (TCR) Recognition: Crucially, will the circulating T-cells recognize the peptide-MHC complex as a threat?
Many high-affinity binders fail to elicit an immune response in vivo because they lack true immunogenicity or are subject to central tolerance. Therefore, prioritizing the right neoantigens means the difference between a vaccine that successfully shrinks a tumor and one that merely provokes a harmless, silent biological event.
The Intersection: Designing the Optimal mRNA Construct
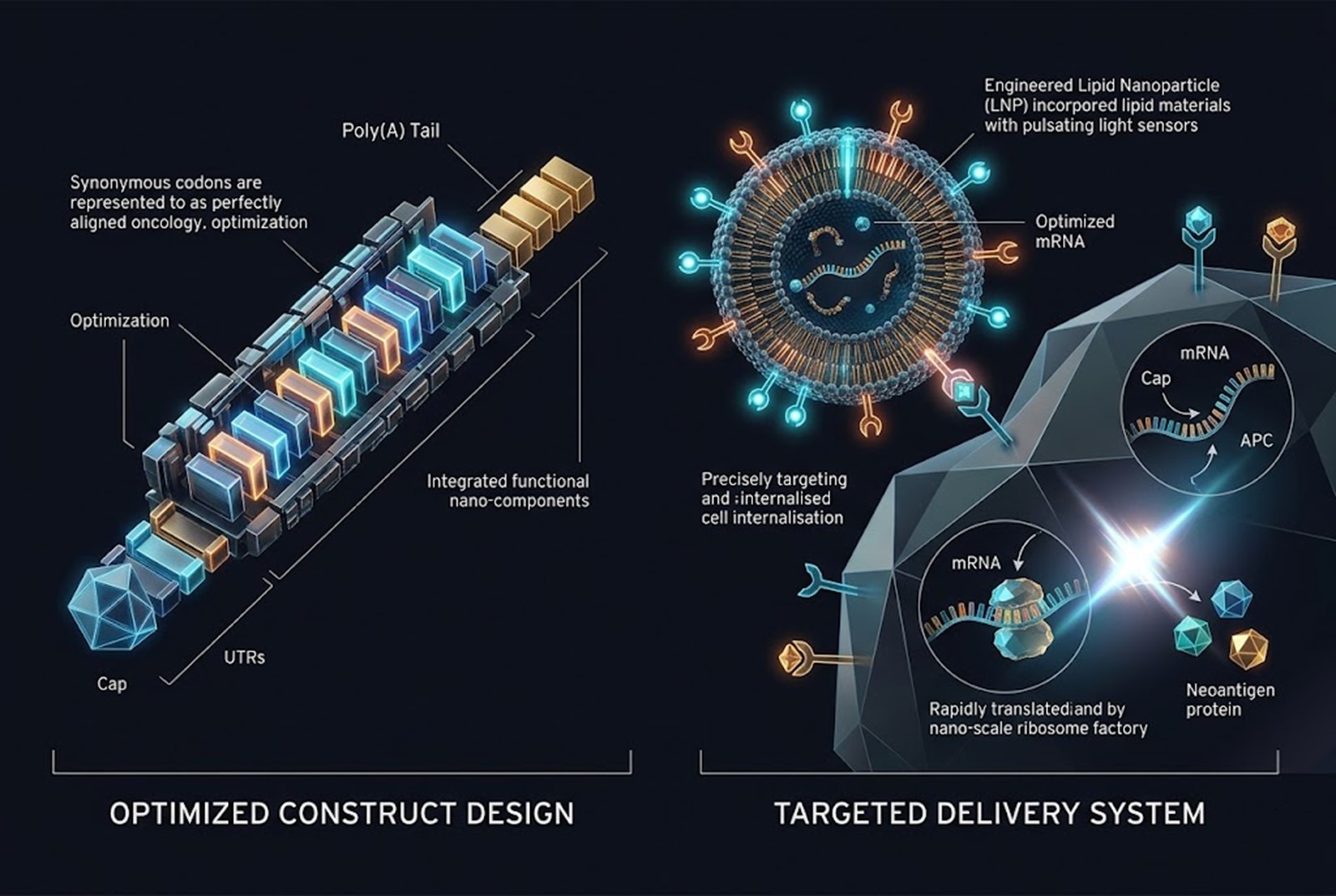
Once the top candidates have survived the rigorous neoantigen prioritization process, the focus shifts to translating these specific peptide sequences into an optimized mRNA construct. This is the critical junction where prioritization meets deliverability. The raw genetic sequence cannot simply be injected; it must be extensively engineered to survive the harsh journey into the patient’s cells and maximize protein expression.
Effective mRNA vaccine design involves intricate modifications to the transcript itself:
-
5′ Cap and 3′ Poly(A) Tail: These structures are critical for protecting the mRNA from exonuclease degradation in the cytoplasm and facilitating ribosome recruitment.
-
Untranslated Regions (UTRs): The 5′ and 3′ UTRs must be carefully selected or synthetically designed to maximize translational efficiency and the intracellular half-life of the molecule.
-
Codon Optimization: Synonymous codon optimization is employed to match the tRNA pool of the target cells, speeding up translation without altering the final, carefully prioritized amino acid sequence of the neoantigen.
Importantly, the secondary structural stability of the mRNA molecule directly impacts its deliverability. Complex secondary structures within the coding sequence can impede ribosomes, while unfavorable folding dynamics can interfere with encapsulation into delivery vehicles. Thus, the bioinformatics algorithms used for neoantigen prioritization must increasingly work in tandem with RNA folding prediction models to ensure the chosen sequences can be physically formulated.
Overcoming the Deliverability Hurdle: Beyond the Sequence
A perfectly prioritized neoantigen encoded in a highly optimized mRNA transcript is clinically useless if it degrades in the bloodstream or fails to enter the correct immune cells. Naked mRNA is highly susceptible to rapid degradation by ubiquitous extracellular RNases and is too large and negatively charged to passively cross the lipid bilayer of a cell membrane. Therefore, physical deliverability is the ultimate gatekeeper of therapeutic success.
Lipid Nanoparticles (LNPs) are currently the gold standard for mRNA delivery. These intricate spheres of synthetic lipids encapsulate the mRNA, protecting it from degradation and facilitating endosomal escape once internalized by a target cell. In oncology mRNA vaccine design, the delivery vehicle must do more than just enter any random cell; it must preferentially target Antigen Presenting Cells (APCs), particularly dendritic cells, which are the biological sentinels responsible for initiating the required CD8+ T-cell response.
Achieving this targeted deliverability requires modifying the LNP composition—adjusting the ratios of ionizable lipids, structural lipids, cholesterol, and PEGylated lipids—or adding specific targeting ligands to the nanoparticle surface. Furthermore, the formulation must balance efficient cellular uptake with acceptable systemic tolerability, avoiding excessive inflammation or hepatotoxicity that could limit the safe therapeutic dose.
AI: The Unifying Force in Vaccine Engineering
The sheer volume of variables involved in integrating neoantigen prioritization with LNP deliverability considerations far exceeds the capacity of traditional trial-and-error methodologies. Consequently, Artificial Intelligence (AI) is rapidly becoming the indispensable core of modern oncology mRNA vaccine design.
Deep learning models are now capable of analyzing massive, multimodal datasets—from patient genomic profiles and TCR repertoires to lipid chemical structures and RNA folding dynamics. By utilizing AI, researchers can predict not only which neoantigens will be most immunogenic but also how the corresponding mRNA sequence should be structured for optimal LNP encapsulation and maximum expression in vivo. This holistic, AI-driven approach significantly reduces the time, cost, and failure rates associated with pre-clinical development, moving the biopharma industry closer to the ultimate goal of rapid, on-demand personalized cancer therapeutics.
Accelerate Your Research with Creative Biolabs
At Creative Biolabs, we leverage advanced artificial intelligence to streamline and optimize your biopharmaceutical research. If you are developing the next generation of therapies, explore our specialized AI-driven services designed to support your pipeline:
